WHAT´S HOT
BHB PRESENTS THE NEW BFE BIOKIT, A REVOLUTION IN THE FACE MASKS TESTING
BHB is pleased to announce the commercialization in Portugal of the first certified industrial solution for the testing of face masks according to EN 14683:2019.
The BFE Biokit was developed by the Italian manufacturer TCR Tecora at the request of the Italian Army and the Italian National Council of Health, with the aim of reducing the testing time required to validate the conformity of face masks.
Already implemented in Italy, it has proved to be an effective and low-cost alternative to traditional methods and is now being produced and marketed throughout all Europe.
With a wide range of applications, from pharmaceuticals to textiles, it is a simple, robust system that combines the highest precision with the low cost of operation and maintenance.
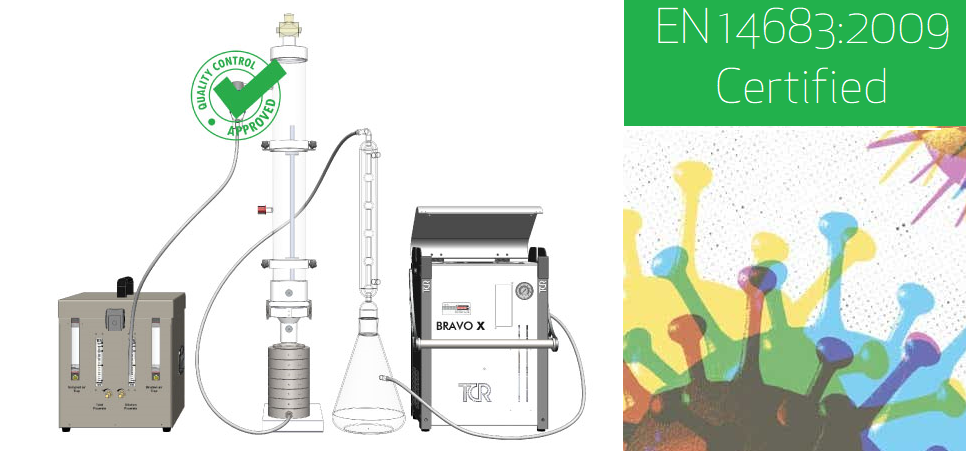
Above - TCR Tecora BFE BioKit pneumatic diagram detail according to the EN 14683:2019 standard (Annex B).
BIOAEROSOL COMPLETE SAMPLING SYSTEM
Bioaerosol is a component of Particulate Matter (PM) in the atmosphere and consists of airborne particles that have a biological origin. Bioaerosol has a very varied composition, including:
- Microorganisms (viruses, bacteria, fungi, and their spores, algae, protozoa);
- Pollen;
- Fragments of animals, insects, plants;
- Derived substances (e.g. toxins and allergens) produced by any living species.
The study of bioaerosols involves many fields of application, from Allergology to biological terrorism. In fact, the study of the microbial content of the air has become increasingly significant in recent years when the need for “contamination-free” environments has become more evident.
Knowing the dimensional distribution of bioaerosol allows us to evaluate its aerodynamic behavior in the atmosphere (air suspension time, transport phenomena and deposition) and its potential effects on human health (deposition in the different sections of the respiratory system).
In the BFE Biokit sampling system, bioaerosol is sampled as a function of it dimension in a multi-stage impactor. In this way, bioaerosol is collected to an impactor surface consisting of a membrane, grease-coated plate or biological collection plate and is studied by different analytical techniques (microscopic investigations, immunological, biological and chemical tests, culture techniques for living cells).

Above - Details of several bioaerosols (algae, Fungus, bacteria, virus and spores).
MULTI-STAGE IMPACTOR
From the point of view of respiratory deposition, the human respiratory system acts as an aerodynamic classifier of airborne particles. The multi-stage impactor, based on the principle of inertial particle impaction, simulates the human respiratory tract (extrathoracic, tracheobronchial, alveolar).
The micro-holes in each of the 6 planes of the impactor act as holes that, depending on the diameter and the impact distance, allow capturing a dimensional range of particles, with an impact curve of specific and characteristic efficiency.
The special design of the multi-stage impactor ensures the chain deposition of particles that are suspended in ambient air by classes.
To mitigate the known "bounce effect" common to impactors, the impactor uses a petri dish with an agar solution, preventing the particles from re-joining or suspending.
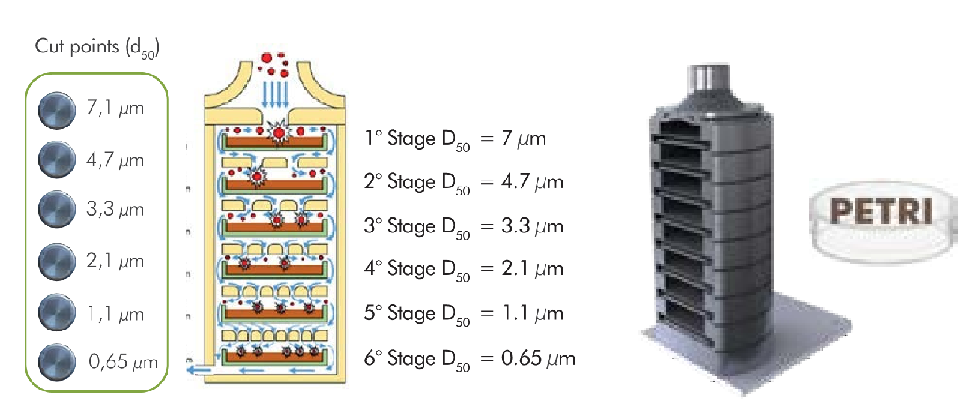
Above - Detail of the operating principle of the impactor (NIOSH Manual of Analytical Methods - Sampling and characterization of Bioaerosol).
PROCESS FOR THE CERTIFICATION OF BREATHING MASKS
In order for a breathing mask to be tested and validated according to European standards and to obtain a declaration of conformity, it is necessary to carry out several tests.
Standard EN 14683:2019, harmonized under the Medical Provisions Directive, is intended for masks for clinical use by classifying them in different types (I, II and IIR) according to their characteristics, namely: bacterial filtration efficiency, differential pressure (permeability of the mask to air), splash resistance and microbial cleansing (bioburden).
The bacterial filtration efficiency test (called BFE) and the air permeability level test (called differential pressure) are performed automatically by the BFE Biokit, using proprietary software, issuing simple reports that include all necessary points.

Above - Detail of the complete solution - Nebulizer, Impactor, Aerosol Chamber, Condensate Extractor and Flow Sampler.


